The feasibility of a high frequency home-based therapy tailored to GMFCS type 4 and 5 patients was explored via a non-randomized prospective cohort treatment study.
To date, 367 American and South American cerebral palsy type 4 and 5 patients participated. Inclusion criteria was limited to patients over 5 years old, not having undergone surgery over the course of the study, and having at least a 3 year follow up. This left 100 patients having taken part in the study.
At baseline and every 6 months, parents of CP patients were provided a 3 day lesson by experienced physical therapists teaching a safe and simple home-based manual therapy with focus to provide cyclic motion strain to the weaken midsection of their child. Stimulation guidelines (pressure magnitude and frequency) were instructed and monitored for consistency using a custom force gauge integrated into the therapy. Therapy was encouraged for at least 30 minutes 5 times a week. CPCHILD questionnaires were collected at baseline and bi-annually for 3 years by the physical therapists. Additionally, measures of intra-abdominal pressure (force of 1 compressive inch), seated spinal stability (minimal support for 10 seconds of independent sitting), and the GMFCS classification were taken initially and bi-annually for 2 years. Study directives were approved by independent ethical committees. Non-parametric Wilcoxon tests were utilized to perform data analyses.
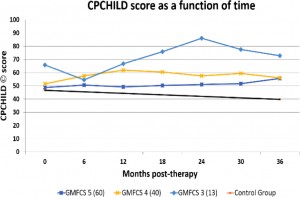
Baseline CPCHILD scores agreed with published mean values per GMFCS class. To date, CPCHILD scores of GMFCS 4 patients improved by 8% from 51.55 to 56.02 and GMFCS 5 improved by 14% from 48.74 to 55.54 after 36 months of home therapy (p<0.05). Measures of intra-abdominal pressure improved from 3.23 to 4.46 lbs/inch of compression and, correspondingly, seated spinal stability improved from 2.3 to 3.1 after 36 months (p<0.05). Average GMFCS scores improved from 5 to 4 while types GMFCS 4 remained the same after 24 months.
Results suggest a high frequency home based therapy for CP patients to be a feasible platform for the improved health and wellbeing of severely affected cerebral palsy patients GMFCS types 4 and 5.
Medical relevance of alternative treatments for cerebral palsy
ABR is considered an alternative treatment which, by definition, refers to a treatment avenue that seeks to improve disease or disability that is not yet approved by health authorities. In no manner does this connote or entail that the treatment is unproven. Such fallacious perceptions are fueled by the fact that the field of alternate therapies is littered with self-proclaimed panaceas whose ambiguous promises are at best questionable. Unfortunately, the grim realities of developmental disabilities provide no easy answers and thus drive this continuous search for new and better treatments.
However, it should be considered that there exist alternative therapies that distinguish themselves by stringently adhering to the scientific methods that have previously paved the path of today’s established care methods.
Study on the efficiency of ABR Therapy
Score improved by 7 points.
GMFCS type 4 patients improved their CPCHILD scores by 5 points (8%, p<0.05) over initial measures after 36 months of home therapy. Correspondingly, GMFCS type 5 patients improved their CPCHILD scores by 7 points (14%, p<0.05). Perhaps most importantly results are also clinically significant.
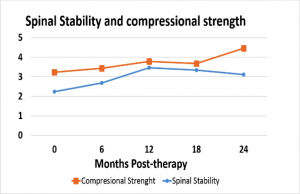 Intra-abdominal pressure and spinal stability
Intra-abdominal pressure and spinal stability
The average initial force required for abdominal compression of 1 inch was 3.2 lbs and after 2 years was improved significantly to 4.6 lbs (p<0.05). A higher measure is analogous to a stronger core. Furthermore, the baseline measure of spinal stability was 2.2 and improved consecutively to 3.1 after 2 years (p<0.05). For both test 70 patients with cerebral palsy took part in the study.
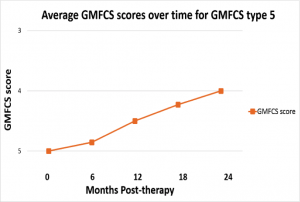 Measures of the GMFCS classification
Measures of the GMFCS classification
Progression from level 4 to level 5
The average baseline measure was a GMFCS classification of 5 suggesting the majority of the patients lied within the more limited individuals. The average measure after 2 years was reduced (a reduction suggest improved function) in a consistent manner from 5 to 4.